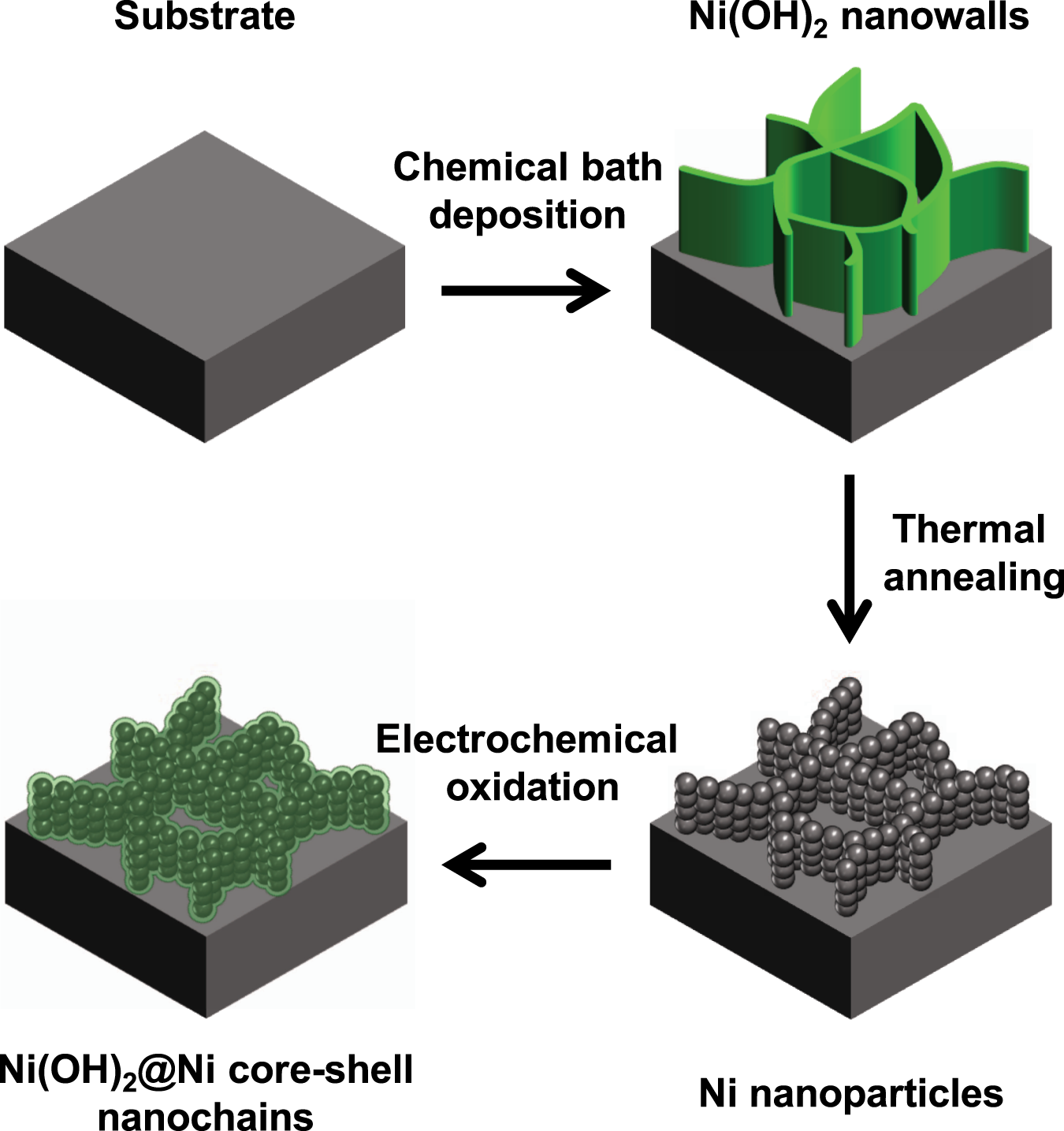
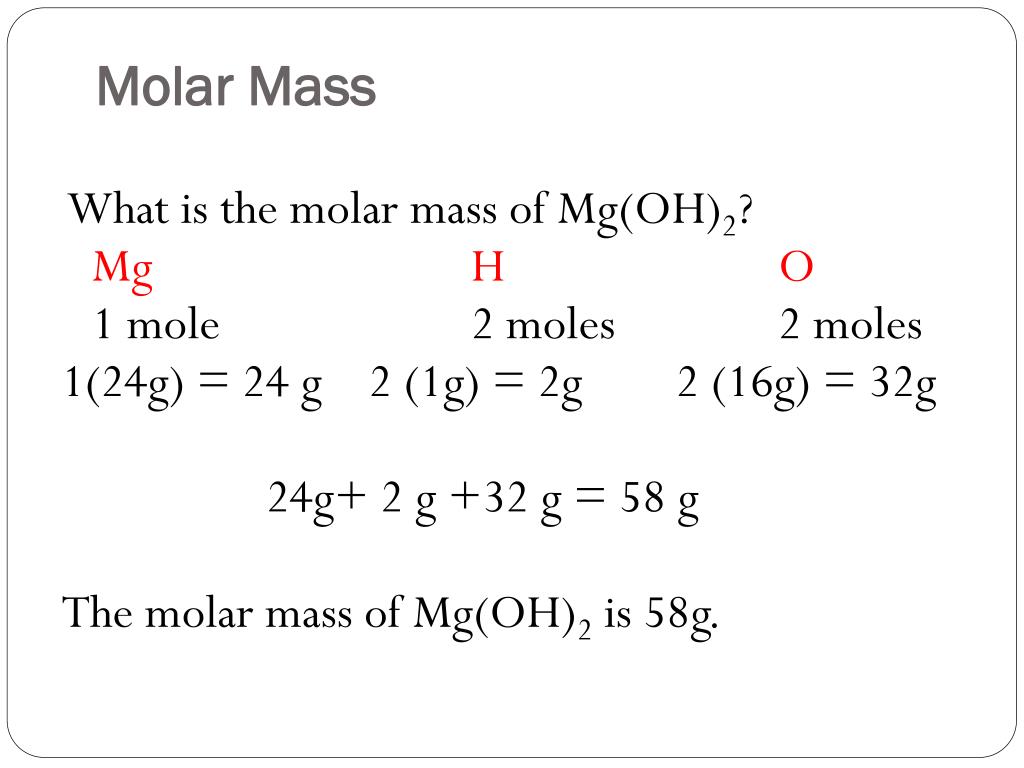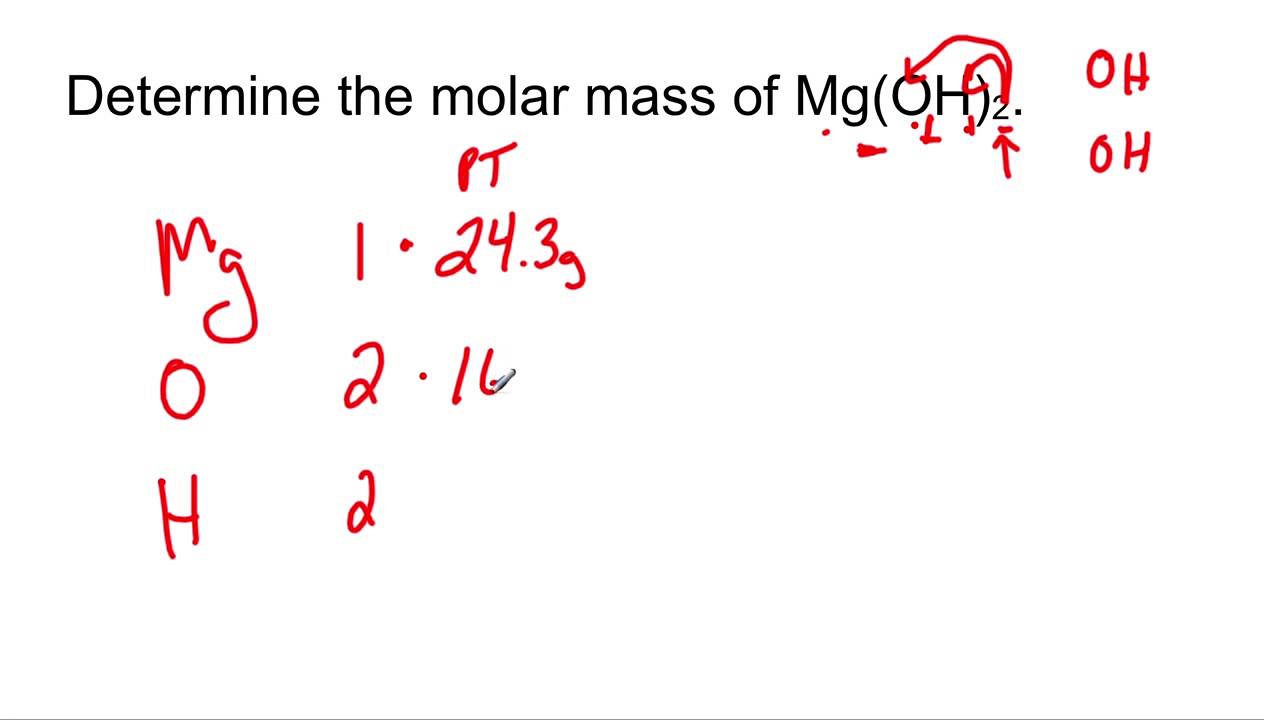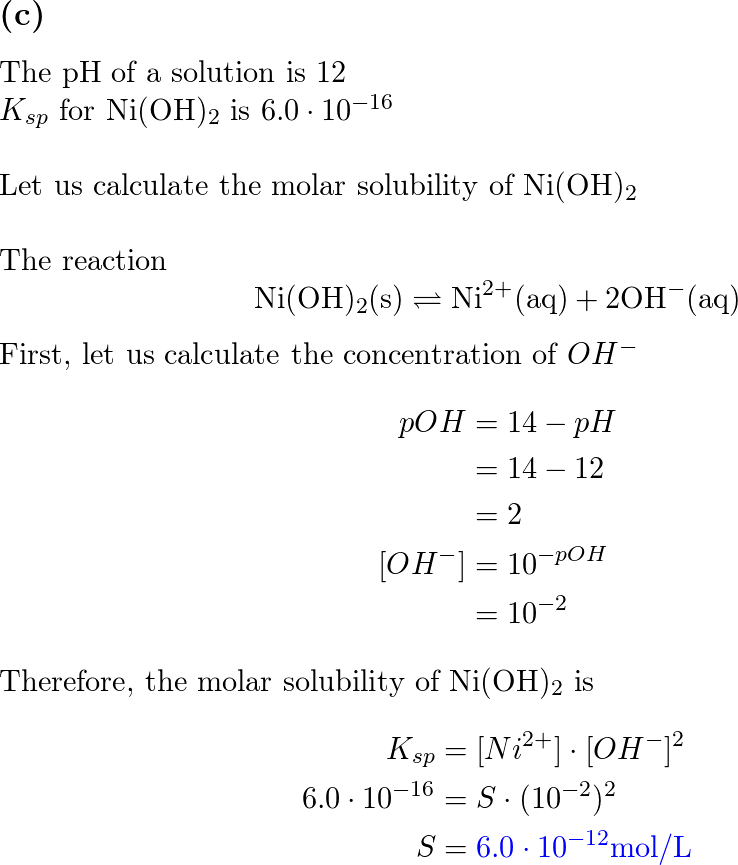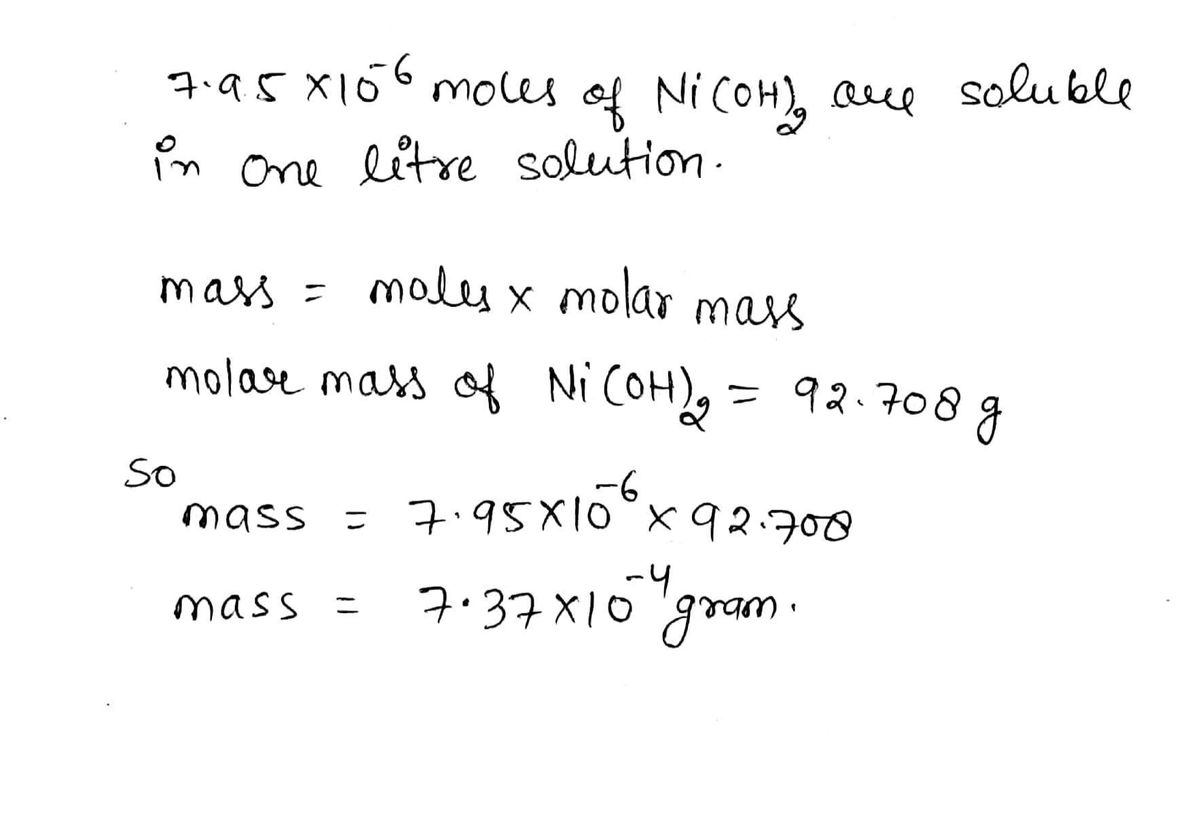Ni(OH)2@Ni core-shell nanochains as low-cost high-rate performance electrode for energy storage applications | Scientific Reports

Realizing Two-Electron Transfer in Ni(OH)2 Nanosheets for Energy Storage | Journal of the American Chemical Society

Calculate the molar solubility of Ni(OH)2 in 0.10M NaOH. The ionic product of Ni(OH)2 is..... - YouTube

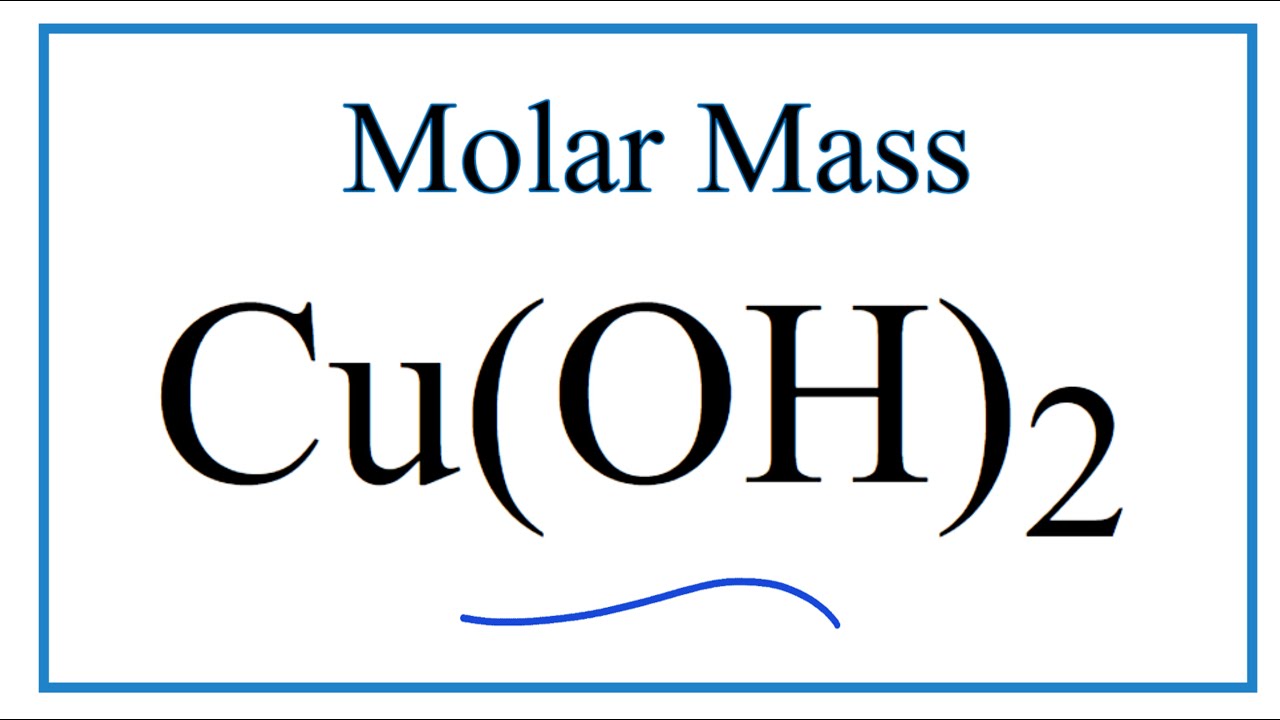
MOLAR MASS The molar mass is the mass, in grams, of one mole (6.02 1023 particles) of an element (ion), a covalent molecule or a formula unit. molar. - ppt download

Ni(OH)2 Nanoplates Grown on Graphene as Advanced Electrochemical Pseudocapacitor Materials | Journal of the American Chemical Society
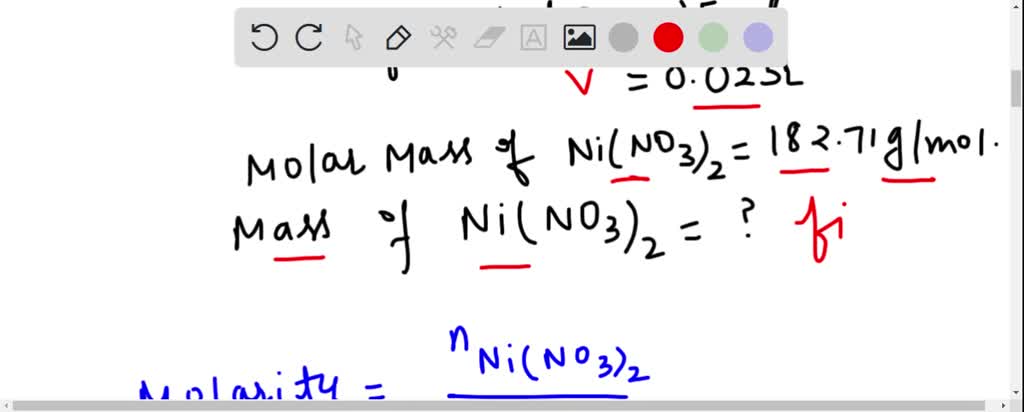
SOLVED: What is the mass of nickel(II) nitrate (182.71 g/mol) Ni(NO3)2 dissolved in 25.0 mL of 0.100 M solution? 4.00810.457 g 45.7 0.250 B 457 g

A mixture of 100mmol of Ca(OH)2 and 2g of sodium sulphate was dissolved in water and the volume was made up to 100mL. The mass of calcium sulphate formed and the concentration

SOLVED: The molar solubility of nickel(II) hydroxide (Ni(OH)2) is 2.8 x 10-6 mol/L in pure water at 25°C. What is the molar solubility of nickel(II) hydroxide in 0.050 M NaOH at 25°C? (

Ni(OH)2 Nanoplates Grown on Graphene as Advanced Electrochemical Pseudocapacitor Materials | Journal of the American Chemical Society
Unanswered Question 5You AnsweredCorrect Answer0/1 pts554. mL of 0.652 M KOH(aq) is mixed with 518. mL - brainly.com